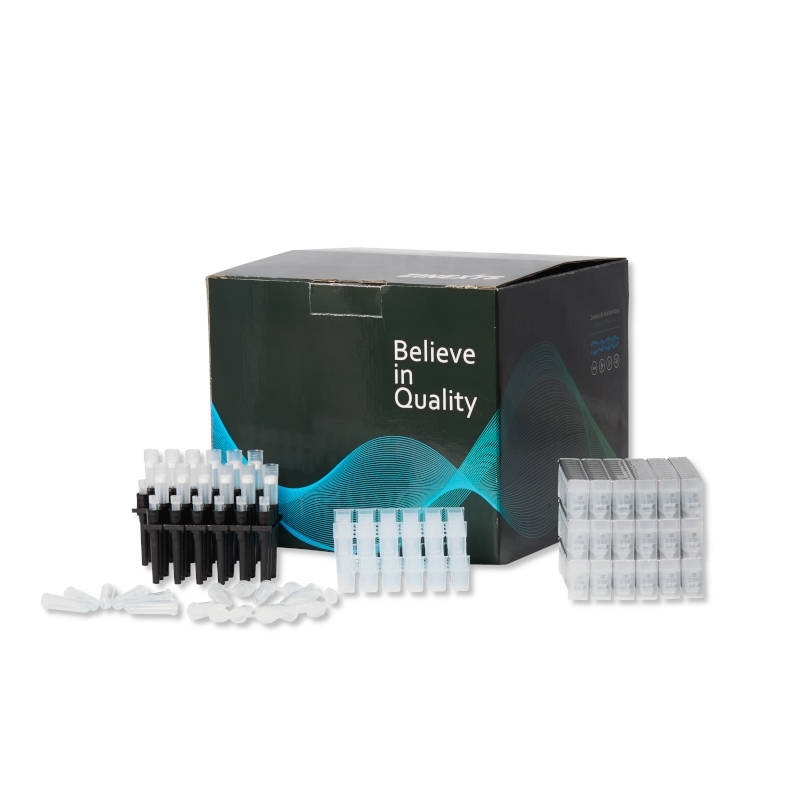
MagPurix® Reagent Kits are designed to provide the user the highest extraction quality with minimum human interaction. These optimized protocols together with an easy-to-use user interface allow laboratory technicans utilizing their time much better. All reagent kits are available in pre-packed environmentally friendly boxes containing all the components for 48 extractions : separable reagent cartridges, polygon reaction chambers®, tip holders, single sample tubes, elution tubes, filter tips and piercing pins, as well as additional specific buffer and optional RNA carrier.
The uniquely designed magnetic bead processing chamber provides the highest quality results and efficiently removes all inhibitors. Its smart design allows the user to include an Internal Control (IC) before the isolation procedure's startup.
The kit uses ZiBeads® unique patented magnetic technology that delivers high quality nucleic acids in consistently high yields, in combination with MagPurix® series automated instruments.
The isolated viral DNA and RNA are of exceptional purity and are suitable for PCR and real-time PCR, genotyping, sequencing (NGS), oncology or pharmacogenomics research and other typical application analysis. It is also compatible for direct PCR analysis.
The kit is supposed to be used on the MagPurix®12/24 series, on the MagPurix®12/24 EVO and on the MagPurix®N.E.O., allowing the purification of variable number of samples at once.
MagPurix® Extraction Kits are supplied in ready-to-use, prefilled reagent cartridges and specially designed plastic consumables, which ensures speed and convenience in sample preparation and instrument setup.